Five passengers lost their lives and forty more were injured, and I know I speak for all the House when I say that our thoughts are with the injured and the relatives of the deceased passengers.

– Dear colleagues, on Friday last week, a train was derailed in Bavaria. Откриване на заседанието Apertura de la sesión Zahájení denního zasedání Åbning af mødet Eröffnung der Sitzung Istungi algus Έναρξη της συνεδρίασης Opening of the sitting Ouverture de la séance Apertura della seduta Sēdes atklāšana Posėdžio pradžia Az ülés megnyitása Ftuħ tas-seduta Opening van de vergadering Otwarcie posiedzenia Abertura da sessão Deschiderea ședinței Otvorenie rokovania Otvoritev seje Istunnon avaaminen Öppnande av sammanträdet Otvaranje dnevne sjednice Oscailt an tsuí (The sitting opened at 17.02) Изявление на председателството Declaración de la presidencia Prohlášení předsednictví Erklæring fra formanden Erklärung der Präsidentschaft Presidentuuri avaldus Δήλωση της Προεδρίας Statement by the President Déclaration de la Présidence Dichiarazione della Presidenza Priekšsēdētājas paziņojums Pirmininko pareiškimas Az elnök nyilatkozata Dikjarazzjoni tal-Presidenza Verklaring van de Voorzitter Oświadczenie Przewodniczącego Declaração da Presidência Declarația președinției Vyhlásenie Predsedníctva Izjava predsedujoče Puhemiehen julkilausuma Uttalande av talmannen Izjava Predsjedništva Ráiteas ón Uachtarán EN President. – I declare resumed the session of the European Parliament adjourned on Thursday 19 May 2022. KESÄKUUTA 2022 MÅNDAGEN DEN 6 JUNI 2022 PONEDJELJAK 6 LIPNJA 2022 DÉ LUAIN, 6 MEITHEAMH 2022 PRESIDENZA: ROBERTA METSOLA President Възобновяване на сесията Reanudación del período de sesiones Pokračování zasedání Genoptagelse af sessionen Wiederaufnahme der Sitzungsperiode Istungjärgu jätkamine Επανάληψη της συνόδου Resumption of the session Reprise de la session Ripresa della sessione Sesijas atsākšana Sesijos atnaujinimas Az ülésszak folytatása Tkomplija tas-sessjoni Hervatting van de zitting Wznowienie sesji Reinício da sessão Reluarea sesiunii Pokračovanie prerušeného zasadania Nadaljevanje zasedanja Istuntokauden uudelleen avaaminen Återupptagande av sessionen Nastavak zasjedanja Athchromadh ar an seisiún EN President. JÚNIUS 6., HÉTFŐ IT-TNEJN 6 TA' ĠUNJU 2022 MAANDAG 6 JUNI 2022 PONIEDZIAŁEK, 6 CZERWCA 2022 SEGUNDA-FEIRA, 6 DE JUNHO DE 2022 LUNI 6 IUNIE 2022 PONDELOK 6. Armadin (mexidol) solution for injections 50 mg/ml.Cardioviol (Rhizomata cum radicibus Valerianae) 50 ml.Armadin (mexidol) Long tablets with prolonged release 500 mg.Keep out of reach of children at a temperature not exceeding 30 ° C. If you experience an adverse reaction due to the use of the drug, consult your doctor. People with hypersensitivity may have allergic reactions, including rashes, itchy skin, redness of the skin. Given the low toxicity of the drug, the likelihood of poisoning is low even in case of accidental excess of the therapeutic dose. Treatment is continued with the use of Nucleo C.M.F. Forte at a dose of 1 injection once a day. Before administration, the solvent must be transferred into an ampoule with lyophilized powder.Īdults, including elderly patients, are prescribed. Forte, a lyophilisate for solution for injection for intramuscular use. The dosage is set by the doctor individually. Use of the drug during pregnancy or breastfeeding is possible if, in the opinion of the physician, the expected positive effect for the woman outweighs the potential risk to the fetus / child. There are no adequate data from the use of the drug in children. Not established, except for known allergic reactions to certain components of the drug. Facial, trigeminal neuralgia, intercostal neuralgia, lumbago. Treatment of bone and joint neuropathies (sciatica, radiculitis), metabolic (alcoholic, diabetic polyneuropathy), infectious origin (shingles). Forte contains nucleotides: cytidine monophosphate (CMF), uridine triphosphate (UTP) – which are widely used to treat diseases of the central nervous system (CNS).
Solvent: water for injections, sodium chloride.
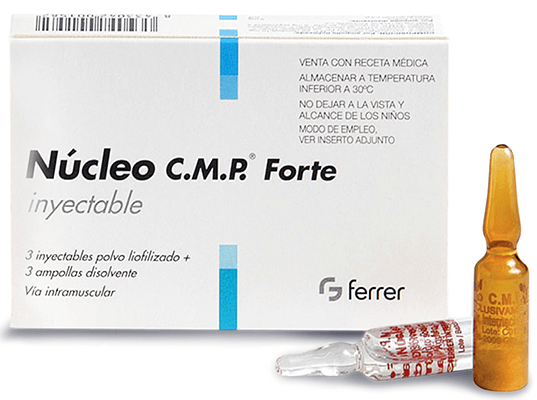
Active substances: sutidine-5-disodium monophosphate (CMP disodium salt), uridine-5-trisodium triphosphate (UTP trisodium salt), uridine-5-disodium diphosphate (UDP disodium salt), uridine-5-disodium monophosphate (UMP disodium salt) ġ ampoule with lyophilisate contains cytidine-5-monophosphate disodium salt (CMF disodium salt) 10 mg, uridine-5-triphosphate trisodium salt (UTF trisodium salt), uridine-5-diphosphate disodium salt of disodium salt, disodium salt monophosphate (UMF disodium salt) only 6 mg (corresponds to 2,660 mg of pure uridine)
